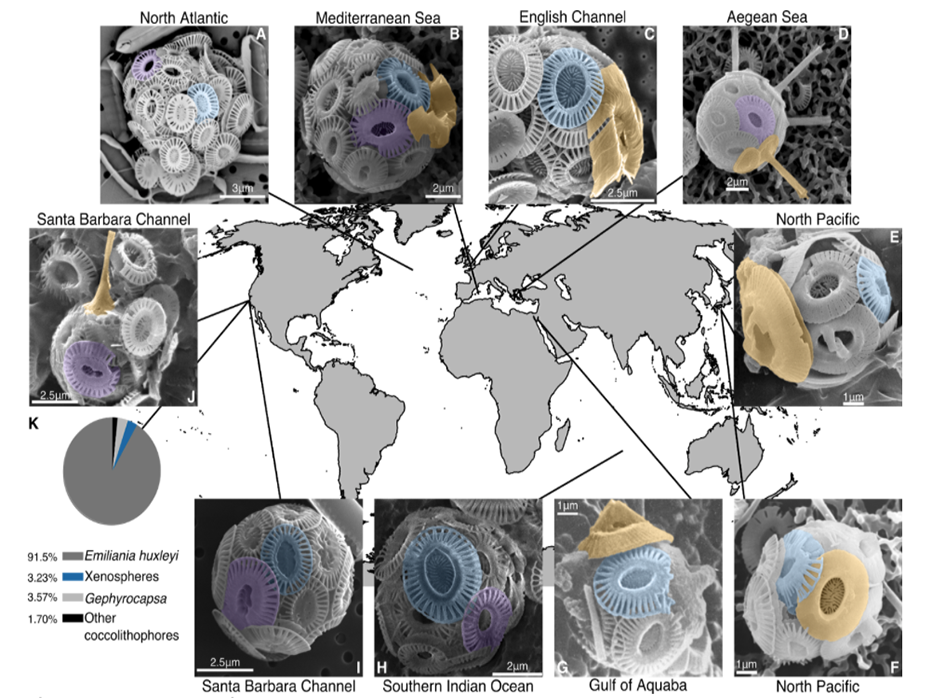
Fig. 1. Xenospheres of coccolithophores are globally distributed across diverse oceanic regions.SEM images of xenospheres composed of: (A) Two different Emiliania huxleyi coccolith morphotypes in the Northeast Atlantic (North Atlantic Virus Infection of Coccolithophores Expedition (NA-VICE;http://www.bco-dmo.org/project/2136); (B) Two different E. huxleyi coccolith morphotypes and a coccolith from another coccolithophore species (possibly Gephyrocapsa oceanica) observed in the Mediterranean Sea [courtesy of Dr. Barbara D’Amario (18)]; (C) E. huxleyi coccoliths and a coccolith from Syracosphaera corolla observed in English Channel [courtesy of Dr. Alison Taylor]; (D) E. huxleyi coccoliths and coccoliths from Rhabdosphaera clavigera observed in the North Aegean Sea [courtesy of Dr. Margarita Dimiza and Odysseas Archontikis (30)]; (E, F) E. huxleyi coccoliths and coccoliths from Gephyrocapsa collected from Uranouchi Bay, Kochi Prefecture, Japan [courtesy of the Electronic Microfossil Image Data Base System (EMIDAS; http://www.emidas.org/) and Dr. Kyoko Hagino (31)]; (G) E. huxleyi coccoliths and a coccolith from G. oceanica in the Gulf of Aquaba (21); (H) Two different E. huxleyi coccolith morphotypes collected during the 4th Indian Southern Ocean Expedition (23); (I) Two different E. huxleyi coccolith morphotypes and (J) E. huxleyi coccoliths and a coccolith from Discosphaeratubiera, both observed in the Santa Barbara Channel [images from Dr. Paul Matson]. (K) Relative distribution of xenospheres (blue) compared to E. huxleyi (grey), G. oceanica (light grey), and other coccolithophore species (black) with uniform, homogenous coccospheres (n=1178). Images from both (I) and (J), as well as data in (K) derive from surface populations across all stations of the 2014 Plumes and Blooms cruise (http://www.oceancolor.ucsb.edu/plumes_and_blooms/). (A-J) Color shading for all SEM images designates E. huxleyi Type A morphotype (blue), other E. huxleyi morphotypes (Type O and Type Overcalcified A; purple), and coccoliths from other coccolithophore species (orange). Scale bars are provided for size reference.
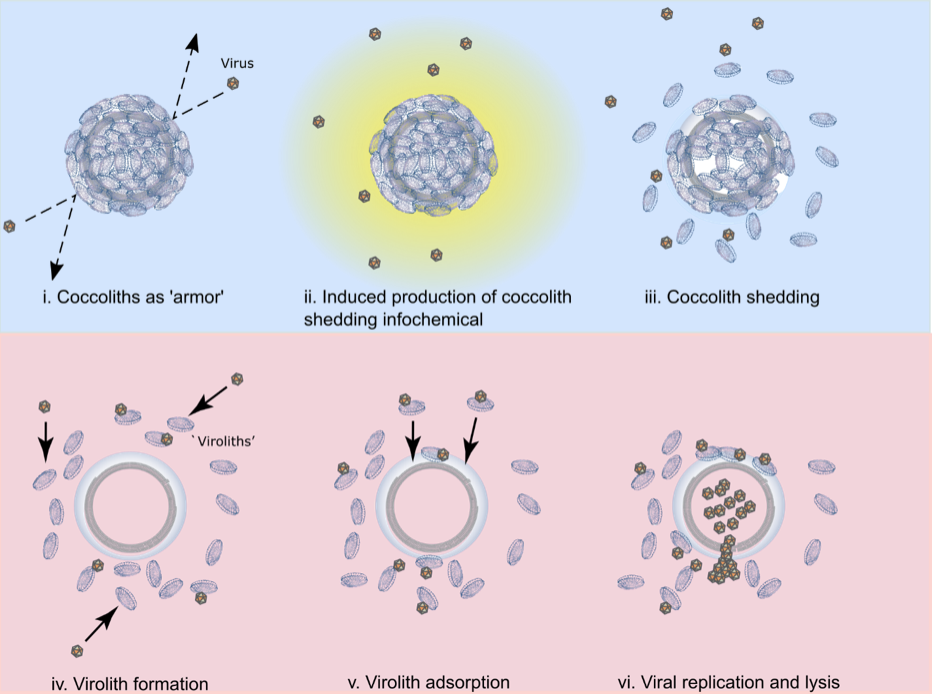
Fig. 2. The multifaceted roles of coccoliths in E. huxleyi-EhV interactions. A conceptual model highlighting the mulitfaceted roles of coccoliths during viral infection. The top section (panels i-iii; shaded blue) represents the beneficial roles of coccoliths to host cells by delaying infection as described in a previous study (5); the bottom section (panels iv-vi; shaded pink) highlights of the antagonistic roles of coccoliths to host cells by facilitating infection via viroliths as described in this study. (i) Coccoliths can help delay infection by preventing viruses from contacting the cell surface; (ii) exposure to viruses induces the production of a host-derived, unidentifed infochemical (yellow) that triggers coccolith shedding; (iii) coccolith shedding occurs prior to cell lysis, increasing the concentration of naked cells and free coccoliths within a population, with the coccoliths adsorbing and removing free viruses from the surrounding milleu; (iv) virus adsorption to free coccoliths leads to the formation of viroliths; (v) virolith adsorption to cells increases the encounter rate of viruses to cells and promotes delivery of infectious EhVs to the cell surface; (vi) viral production and lysis of host cells, in conjuction with virus-induced cocolith shedding, increases the likelihood of further virolith formation.
The dynamic exchange of coccolith biominerals has profound ecological and biogeochemical implications. It puts these biominerals at the heart of interactive arms races between coccolithophore hosts and viruses that toggle cells between life and death. Coccoliths can act as physical protection against viruses but also as a catalyst for death, adsorbing and delivering viruses to the cell for successful infection, analogous to a virulent ‘Trojan Horse’. Persistent coccolith adsorption and exchange fundamentally deviates from prominent paradigms in our field. It has been assumed that coccospheres were produced entirely from within by actively calcifying cells. We show that coccospheres can be made solely of adsorbed coccoliths. Furthermore, demonstration of successful viral infection through biomineral hitchhiking increases host-virus encounters by nearly an order of magnitude (compared to free-floating viruses), critically catalyzing infection and fundamentally altering how we view biomineral-cell-virus interactions in the environment.